
This was a prospective observational study collecting information to evaluate the safety of Tdap vaccine for infants exposed during pregnancy. Skip directly to site content Skip directly to page options Skip directly to A.
Skip directly to site content Skip directly to page options Skip directly to A.
Studies on tdap during pregnancy. Clinicians can find published research about the Tdap vaccine and pregnancy. Skip directly to site content Skip directly to page options Skip directly to A. Pregnant women at 20 weeks 0 days gestation who have not received Tdap during the current pregnancy will be enrolled with prioritized emphasis on enrolling pregnant women who have received Tdap prior to the current pregnancy in order to attempt to address the question of closely-spaced repeated dosing.
Tdap vaccine was received by 658 of pregnant women n 400 in the study. Median gestational age of receipt was 30 weeks. Maternal influenza vaccine receipt infant hepatitis B vaccine receipt provider recommendation of Tdap vaccination and on-site availability of Tdap vaccine were all positively associated with maternal Tdap receipt during pregnancy.
This was a prospective observational study collecting information to evaluate the safety of Tdap vaccine for infants exposed during pregnancy. Infants were followed for between 6 and 12 months after birth with 84 completing 12 months of follow-up. In women during pregnancy and at delivery after vaccination with TdaP in children at birth cord before starting vaccination week 8 one month after primary course of three doses week 20 and before and after the fourth pertussis vaccine dose at month 15 and 16.
Our study validates the current US recommendation to vaccinate with Tdap during pregnancy and suggests that widespread use of Tdap vaccination in pregnancy can result in significant decreases in pertussis particularly in young infants before their first DTaP vaccine dose or who are protected by only 1 dose of DTaP. Becker-Dreps study reviewed more than 675000 pregnancies in the US. From 2010-2014 and analyzed insurance claims data to identify the receipt of Tdap during pregnancy.
National study looking at adverse events following Tdap vaccination during pregnancy and specifically focuses on longitudinal infant outcomes. Women were included in the study if they had received Tdap vaccine between 28 and 38 weeks gesta-tion. They had to have been compliant with routine antenatal care including at least one ultrasound scan.
All pregnant women should get the Tdap vaccine during each pregnancy. The vaccine helps your body make antibodies to protect you from disease. These antibodies pass to your fetus and can protect your newborn until he or she can get the Tdap vaccine at 2 months old.
It is best to get the Tdap vaccine between 27 and 36 weeks of each pregnancy. In this phase 1-2 study initiated prior to the ACIP recommendation to immunize pregnant women with Tdap we evaluated the safety and immunogenicity of Tdap vaccine administered to women in the third trimester of pregnancy and measured placental transfer of maternal pertussis antibodies to the neonate their persistence during the first 2 months of life and their potential effect on infant. Maternal vaccination during pregnancy safe for infants.
New research supports recommendations that pregnant women receive influenza and tetanus diphtheria and acellular pertussis Tdap vaccines. The study found the vaccines are not associated with increased risk of infant hospitalization or death. In an analyses that included more than 300000 births tetanus diphtheria and acellular pertussis Tdap vaccine administration during pregnancy was not.
Receipt of Tdap was not associated with increased risk of hypertensive disorders of pregnancy. Among women who received Tdap at any time during pregnancy 61 percent were diagnosed with. As it stands federal guidelines state that the Tdap vaccine which also guards against diphtheria and pertussis whooping cough is recommended for.
Maternal Tdap vaccination during pregnancy is not associated with autism in the child In this edition of antivaccine Whac-A-Mole Orac discusses a large study that fails to find a link between maternal Tdap vaccination and autism in the baby. No big surprise there. So mothers get your Tdap to protect your baby.
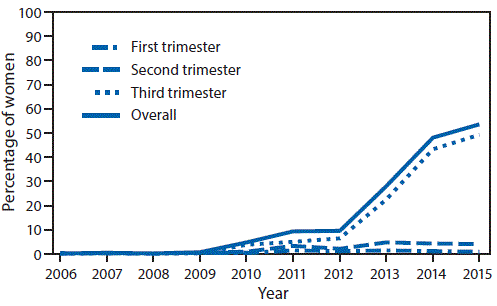
In 2012 the Centers for Disease Control and Prevention CDC recommended that women receive Tdap vaccination with acellular pertussis products during each pregnancy. The American College of Obstetricians and Gynecologists ACOG makes the following recommendations. Obstetric care providers should administer the tetanus toxoid reduced diphtheria toxoid and acellular pertussis Tdap vaccine to all pregnant patients during each pregnancy as early in the 2736-weeks-of-gestation window as possible.